
Helping You and Your Patients Navigate Access to Treatment
For men and postmenopausal women with osteoporosis at high risk for fracture
Not an actual patient.
Support that stands with you
Resources for your office to help start patients on TYMLOS include:
- Prior authorization forms
- Specialty pharmacy network list and intake form
- AACE guidelines
Field Reimbursement Managers (FRMs) help guide:
- Benefits investigations
- Access to prior authorization forms within CoverMyMeds
- The appeals process

TYMLOS SPECIALTY PHARMACY (SP) NETWORK LIST
See a list of non-integrated delivery network SPs that are able to dispense TYMLOS. In addition, SPs affiliated with an integrated delivery network are able to dispense TYMLOS.
Support materials to help get patients started on their journey with TYMLOS.

Starting Your Patients on TYMLOS
Guidance on the steps necessary to prescribe TYMLOS for your patients.
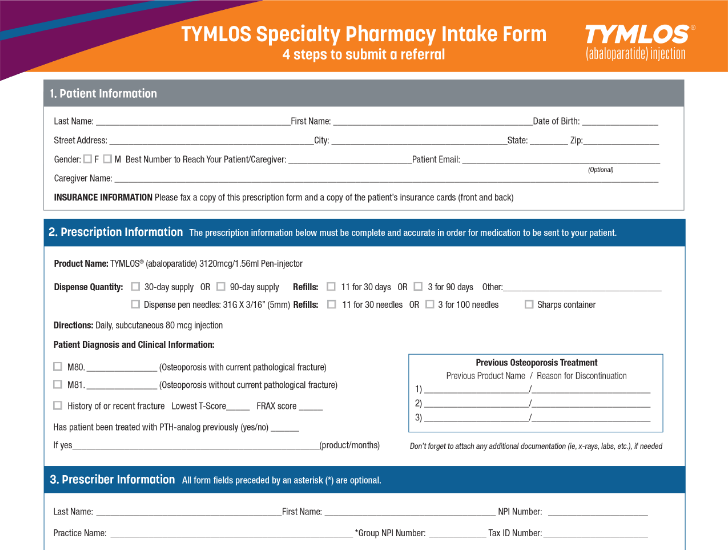
TYMLOS Specialty Pharmacy Intake Form
A form to fill out and fax directly to a specialty pharmacy that dispenses TYMLOS.

Sample of Prior Authorization (PA) Cover Letter
A letter to use as a template when health plans require a PA request form to satisfy their policy requirements to obtain TYMLOS coverage.
Submit prior authorizations electronically through CoverMyMeds in order to streamline the process.
You can call 1-888-452-5017 or visit CoverMyMeds.com to get started.

Sample Letter of Appeal
A letter to use as a template for submitting an appeal when a health plan denies TYMLOS coverage.

Sample Letter of Necessity
A letter to use as a template to support the need for coverage when health plans require more than a letter of appeal.
AFFORDABLE ACCESS FOR MOST MEDICARE PATIENTS
≈30
MILLION
Medicare Part D members have pharmacy benefit insurance for TYMLOS1
$0
OUT-OF-POCKET
$0
DEDUCTIBLE
$0-$12.15 COPAY
Medicare members eligible for “Extra Help” (Low-Income Subsidy or LIS)* will pay no more than a $12.15 copay for TYMLOS when covered3
*A program from the Centers for Medicare & Medicaid Services that may help reduce the financial burden of Medicare Part D premiums and OOP prescription costs for patients with limited income and resources.
SUPPORT FOR PATIENTS
Savings resources for your patients.
We’re here to help your patients find ways to save on TYMLOS.

Savings Card
Eligible,† commercially insured patients can pay as little as $0 a month for their TYMLOS prescription. There is an annual cap on the amount of assistance patients can receive over a 1-year period.

What if your patients are not covered under their INSURANCE plan?
Advise them to call 1‑866‑896‑5674 to learn about a patient assistance program available to eligible patients who cannot afford their medication. Eligibility is based on financial, diagnostic, and insurance criteria.
Empower patients with 1-on-1 virtual injection training from a Clinical Educator.
Once your patients register, one of our Clinical Educators will take them through how to use the TYMLOS pen and self-inject. In addition to our virtual training sessions, your patients will receive our travel-ready injection Welcome Kit and have the option to sign up for the latest TYMLOS updates.
Your patients can register for this unique program at www.TYMLOSsignup.com.
Clinical Educators are also available to provide training for you and your staff on using the TYMLOS pen.

NEED ADDITIONAL INFORMATION OR SAMPLES?
Let us know if you want to request samples or a visit from a TYMLOS representative or field reimbursement manager.
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
References: 1. Data on file. Managed Markets Insight & Technology, LLC. August 1, 2025. 2. Centers for Medicare & Medicaid Services. Final CY 2025 Part D redesign program instructions fact sheet. Published April 1, 2024. Accessed August 1, 2025. https://www.cms.gov/newsroom/fact-sheets/final-cy-2025-part-d-redesign-program-instructions-fact-sheet 3. Centers for Medicare & Medicaid Services. Help with drug costs. Accessed August 1, 2025. https://www.medicare.gov/basics/costs/help/drug-costs
