
For postmenopausal women at high risk of fracture,
WE’RE FUTURE FOCUSED
TYMLOS reduced the risk of fractures with
demonstrated increases in BMD.
For men and postmenopausal women with osteoporosis at high risk for fracture
Not an actual patient.
For postmenopausal women,
A REMODELING ANABOLIC IN ACTION
Safety and efficacy of TYMLOS were evaluated in the ACTIVE Trial.1,2
Impact on Key Skeletal Sites1
TYMLOS demonstrated significant fracture risk reduction vs placebo.1,2
Incidence of New Vertebral Fractures at 18 Months.1*
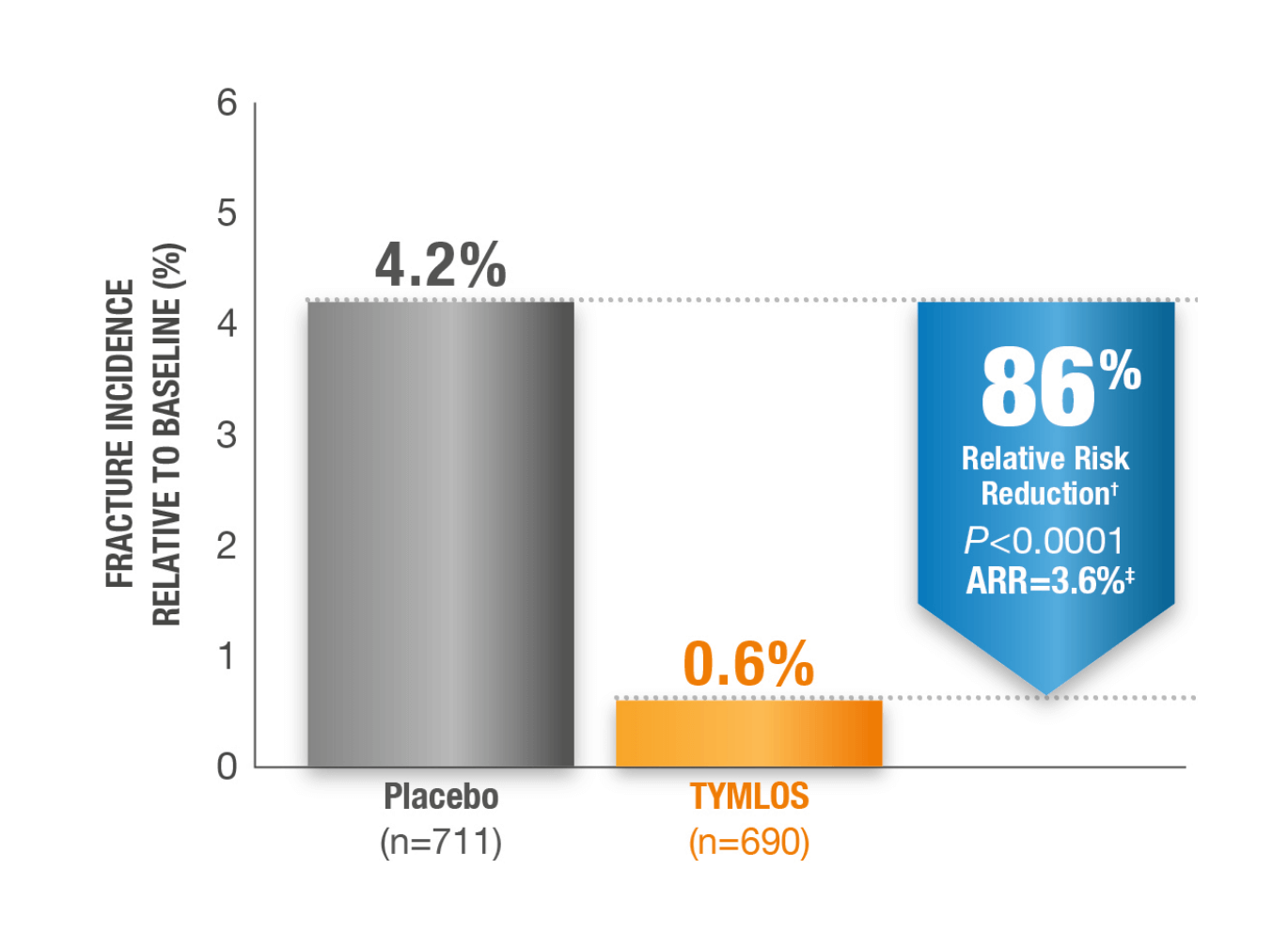

Fracture incidence:
0.6% TYMLOS (n=690) vs
4.2% placebo (n=771)1
*Modified ITT population, which includes patients who had both pretreatment and post treatment spine radiographs.1
†95% CI: 61, 95.1
‡95% CI: 2.1, 5.4.1
Incidence of New Nonvertebral Fractures (Includes Hip Fracture) at 18 Months.1*
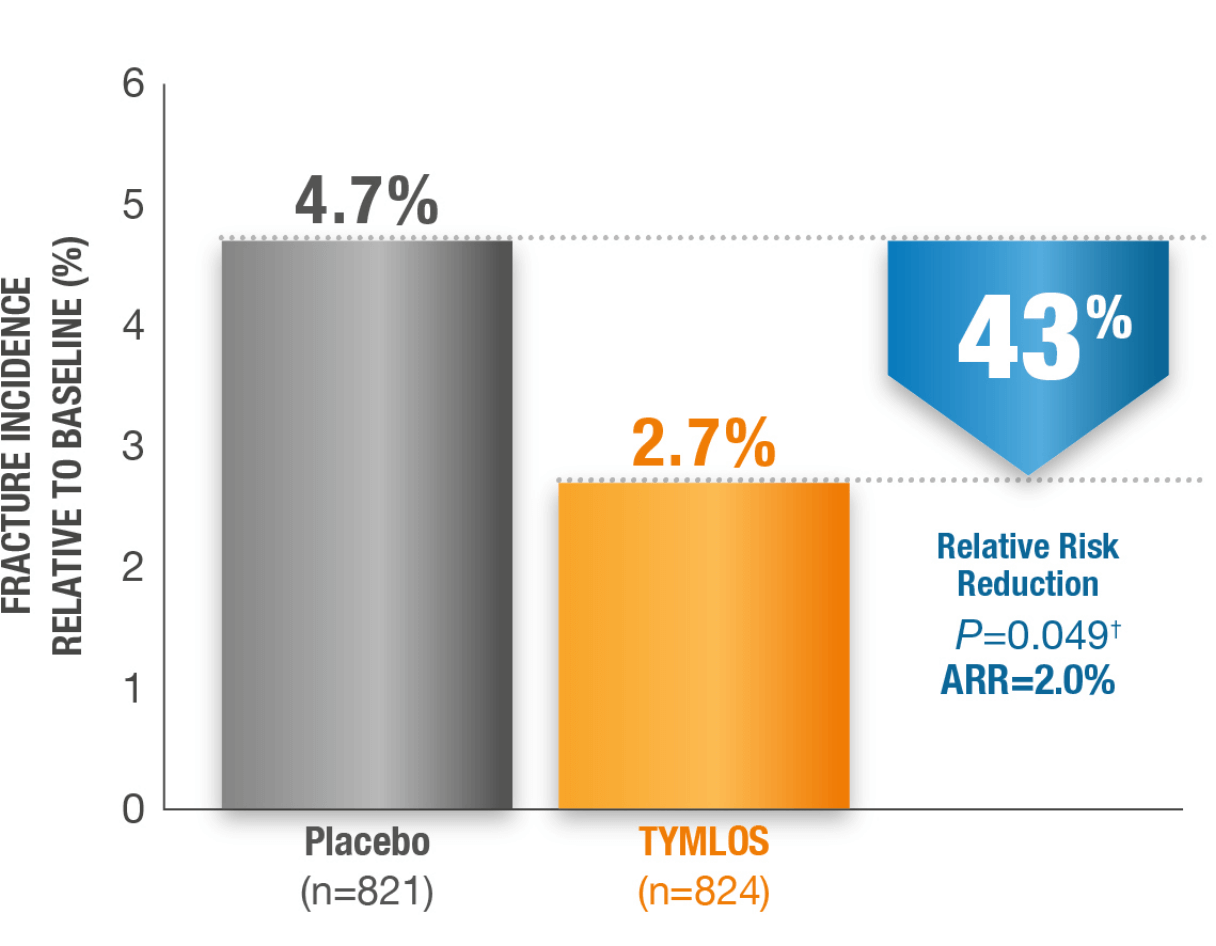
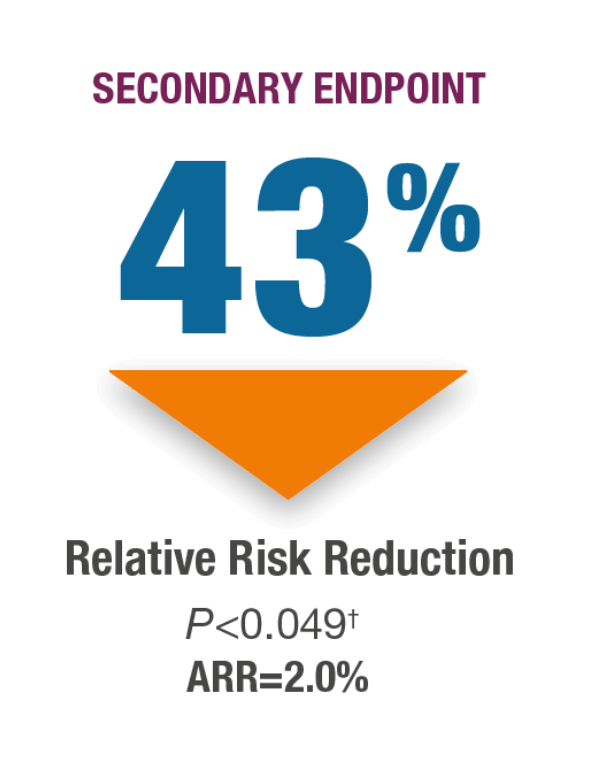
Fracture incidence:
2.7% TYMLOS (n=824) vs
4.7% placebo (n=821)11
*Nonvertebral fractures were measured using the ITT population at 19 months (the entire observational period included 18 months of treatment plus 1 month of follow-up). Nonvertebral fractures excluded fractures of the sternum, patella, toes, fingers, skull, face, and those associated with high trauma.1,2
†P value based on the log-rank test.2
REBUILD BONE AND REDUCE FRACTURE RISK1,2
An exploratory analysis evaluated cumulative incidence of nonvertebral fracture for TYMLOS, teriparatide, and placebo groups at 19 months.2*
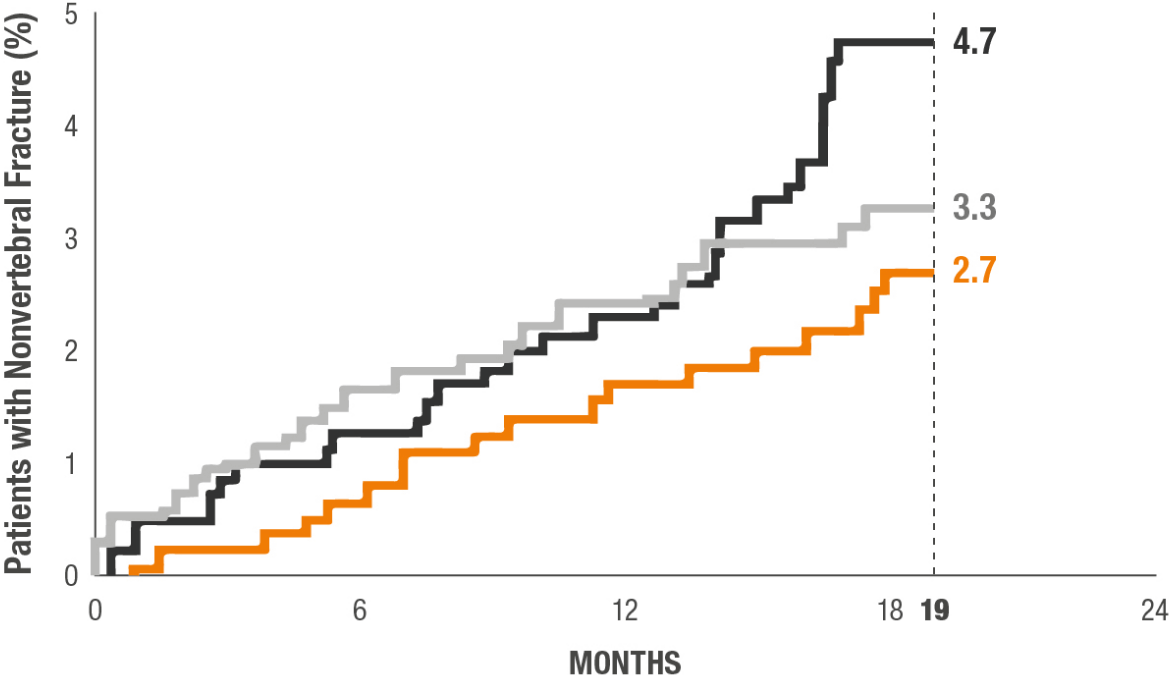
This study was not designed or powered to show, nor can these data be interpreted as evidence of, superiority or noninferiority to teriparatide.
*Nonvertebral fractures were measured using the ITT population at 19 months (the entire observational period included 18 months of treatment plus 1 month of follow-up). Nonvertebral fractures excluded fractures of the sternum, patella, toes, fingers, skull, and face, and those associated with high trauma.2
CI=confidence interval.

TYMLOS (abaloparatide; n=824)
Placebo (n=821)
Teriparatide (open-label active comparator; n=818)
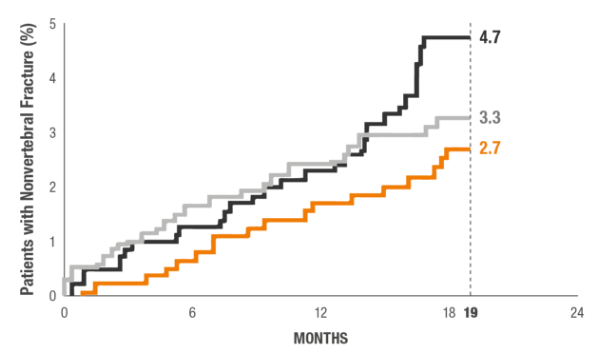
HR (95% CI)
Abaloparatide vs placebo: 0.57 (0.32 to 1.00)
Teriparatide vs placebo: 0.72 (0.42 to 1.22)
Abaloparatide vs teriparatide: 0.79 (0.43 to 1.45)
P value
Abaloparatide vs placebo: 0.049
Teriparatide vs placebo: No significance (NS)
Abaloparatide vs teriparatide: NS
This study was not designed or powered to show, nor can these data be interpreted as evidence of, superiority or noninferiority to teriparatide.
*Nonvertebral fractures were measured using the ITT population at 19 months (the entire observational period included 18 months of treatment plus 1 month of follow-up). Nonvertebral fractures excluded fractures of the sternum, patella, toes, fingers, skull, and face, and those associated with high trauma.2
CI=confidence interval.
TYMLOS helps patients build significant BMD gains vs placebo and protects against bone loss while on treatment.1-3
TYMLOS (abaloparatide; n=824)
Placebo (n=821)
Teriparatide (open-label active comparator; n=818)
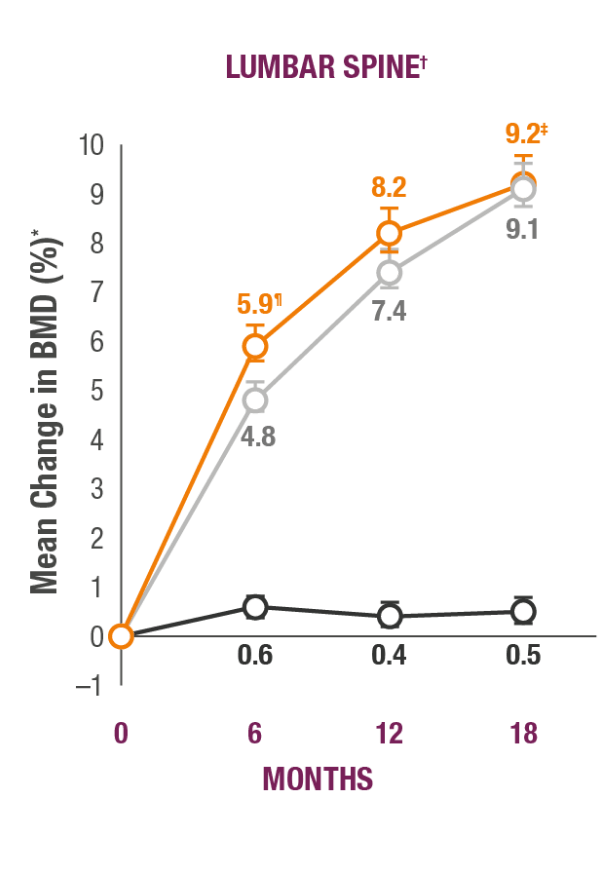
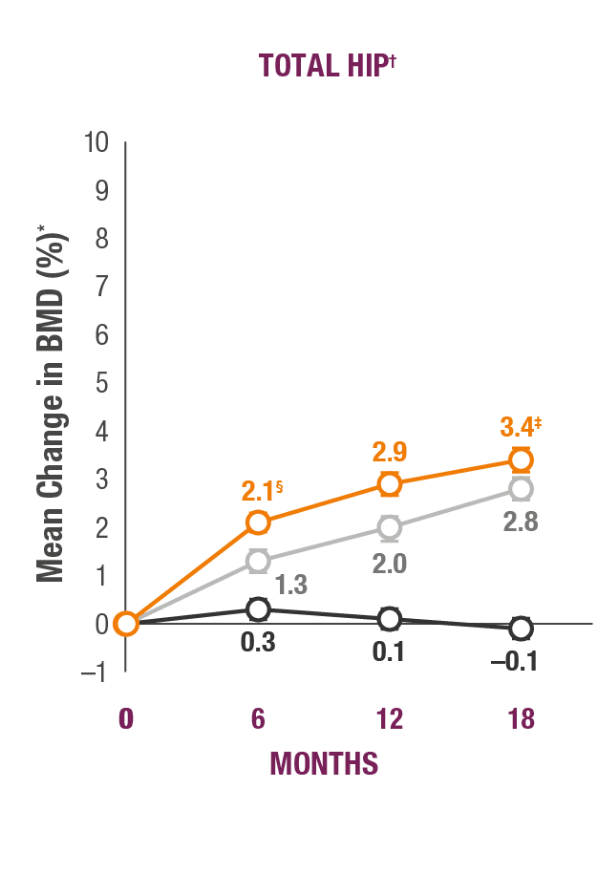
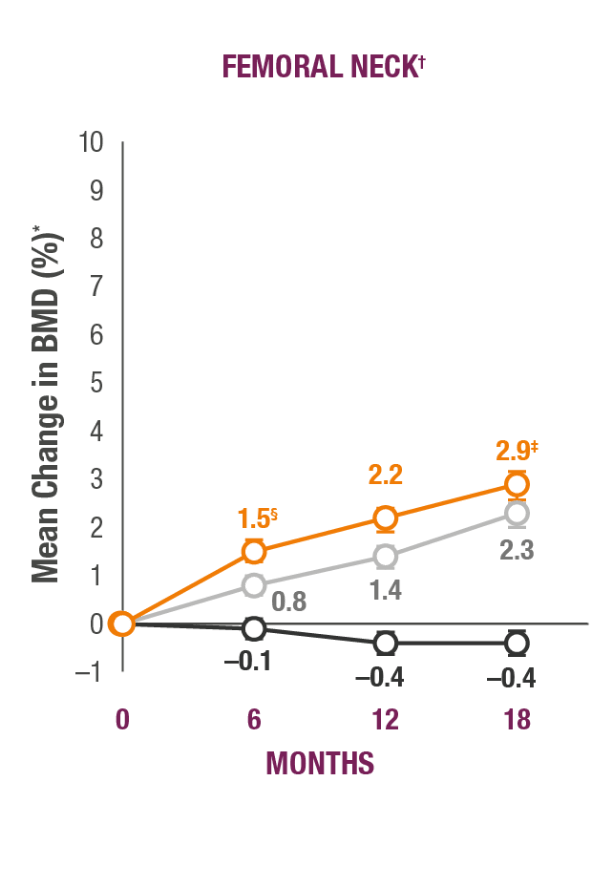
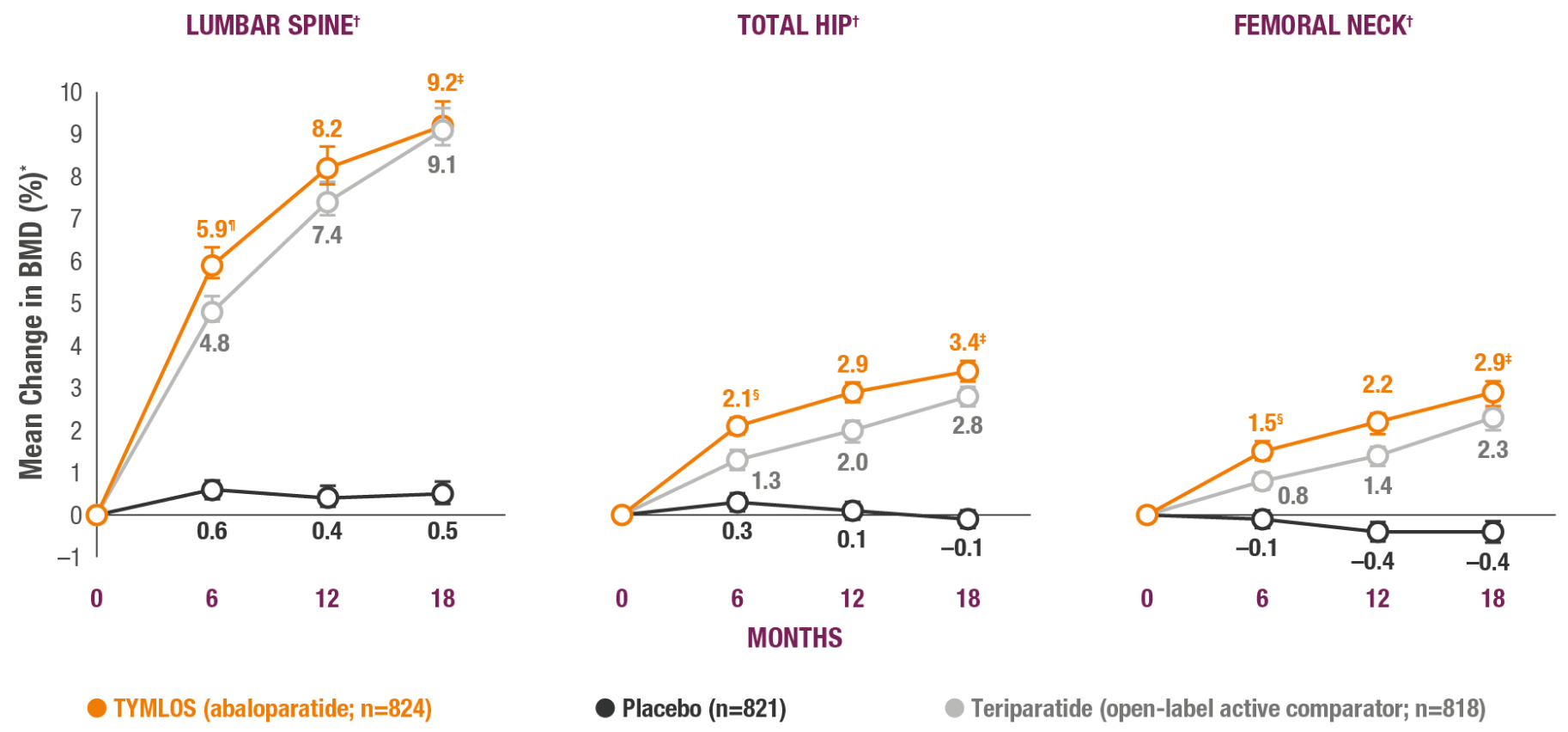
*Results reported in the ITT population, which included patients randomized in the efficacy study; last observation carried forward.1,2
†Error bars indicate 95% confidence intervals.2
‡P<0.0001 vs placebo (secondary endpoint).1
§P<0.0001 vs teriparatide.3
¶BMD changes at lumbar spine vs teriparatide at 6 months was a forced exploratory endpoint due to hierarchical study design.2
From the ACTIVE trial,
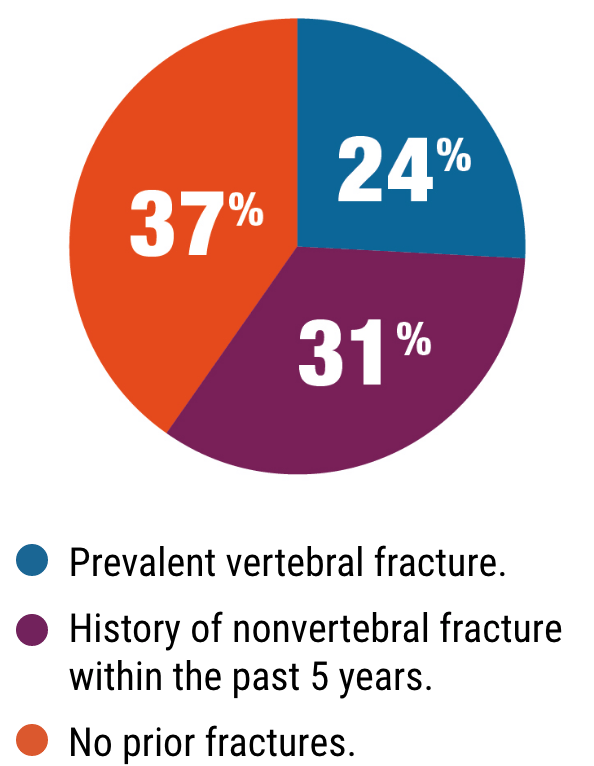
PATIENTS LIKE YOURS2
At baseline, mean T-scores were as follows: Femoral neck (-2.2), total hip (-1.9), lumbar spine (-2.9)2
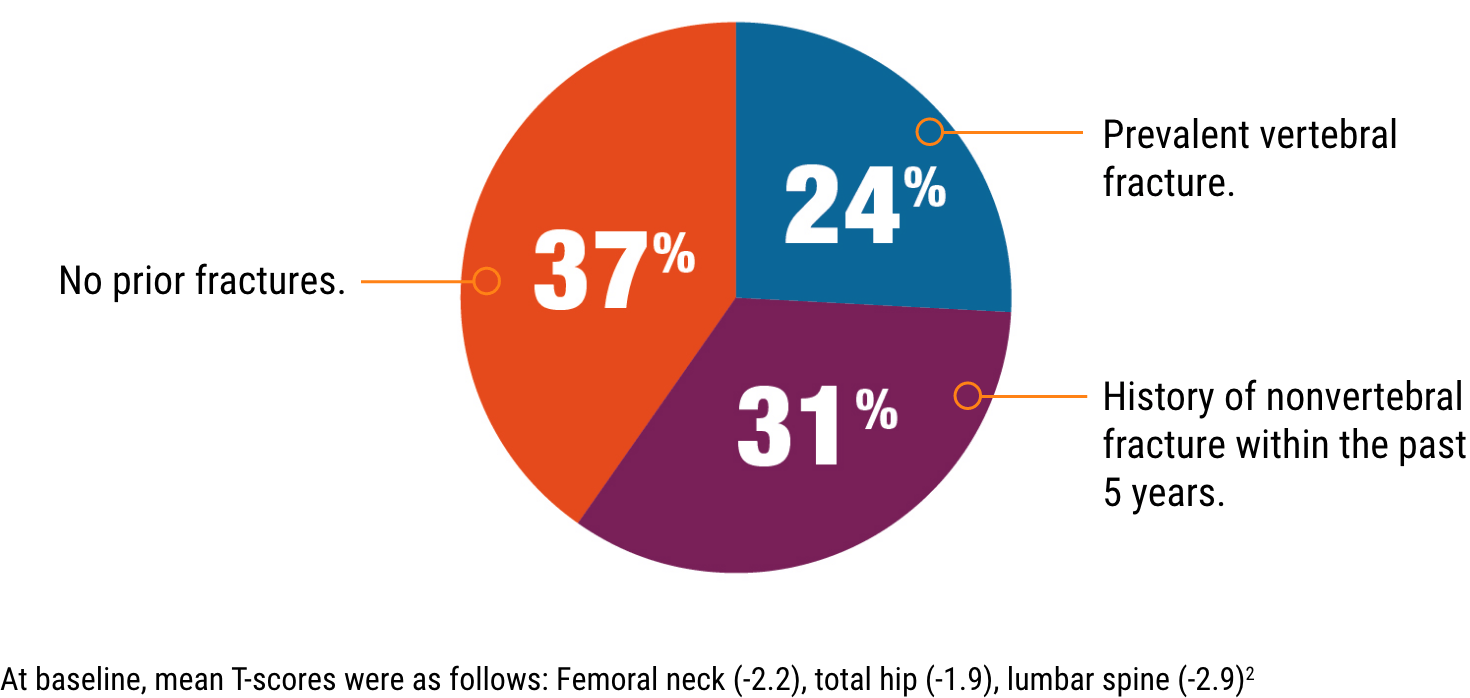
ACTIVE trial participants included patients with:
Type 2 diabetes4
Renal impairment
(up to stage 3 chronic kidney disease)1,5
Varying degrees
of cardiovascular risk1
ESTABLISHED SAFETY OVER TIME1,2,6,7
TYMLOS has a well-established safety profile among women with postmenopausal osteoporosis in the ACTIVE Trial and through 8+ years of real-world use.1,2,6,7*
| MOST COMMON ADVERSE REACTIONS1,2† | TYMLOS (n=822) | Placebo (n=820) |
|---|---|---|
| Hypercalciuria | 11% | 9% |
| Dizziness | 10% | 6% |
| Nausea | 8% | 3% |
| Headache | 8% | 6% |
| Palpitations | 5% | 0.4% |
| Fatigue | 3% | 2% |
| Abdominal pain, upper | 3% | 2% |
| Vertigo | 2% | 2% |
| Hypercalcemia‡ | 3% | 0.1% |
*The safety analysis included an open-label, active comparative arm with teriparatide (N=818).2 This study was not designed to provide head-to-head comparative safety data and cannot be interpreted as evidence of superiority or noninferiority to teriparatide.
†Adverse reactions reported in ≥2% of TYMLOS-treated patients.1
‡Hypercalcemia was a prespecified safety endpoint, defined as albumin-corrected serum calcium of at least 10.7 mg/dL (2.67 mmol/L) at any time point.2
There were no discernible differences in serious adverse events (SAEs) between treatment groups.
Incidence of SAEs:
TYMLOS: 10% vs placebo: 11%1
A majority of patients remained on treatment with TYMLOS.
Discontinuation rates:
TYMLOS: 10% vs placebo: 6%1
SEE HOW TYMLOS OFFERS FLEXIBILITY FOR PATIENTS
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
References: 1. TYMLOS. Prescribing information. Radius Health, Inc. 2. Miller PD, Hattersley G, Riis BJ, et al. Effect of abaloparatide vs placebo on new vertebral fractures in postmenopausal women with osteoporosis: a randomized clinical trial. JAMA. 2016;316(7):722-733. 3. Data on file. Radius Health, Inc. 4. Dhaliwal R, Hans D, Hattersley G, et al. Abaloparatide in postmenopausal women with osteoporosis and type 2 diabetes: a post hoc analysis of the ACTIVE study. JMBR Plus. 2020;4(4):e10346. 5. Bilezikian JP, Hattersley G, Mitlak BH, et al. Abaloparatide in patients with mild or moderate renal impairment: results from the ACTIVE phase 3 trial. Curr Med Res Opin. 2019;35(12):2097-2102. 6. Cosman F, Miller PD, Williams GC, et al. Eighteen months of treatment with subcutaneous abaloparatide followed by 6 months of treatment with alendronate in postmenopausal women with osteoporosis: results of the ACTIVExtend trial. Mayo Clin Proc. 2017:92(2):200-210. 7. Bone HG, Cosman F. Miller PD, et al. ACTIVExtend: 24 months of alendronate after 18 months of abaloparatide or placebo for postmenopausal osteoporosis. J Clin Endocrinol Metab. 2018;103(8):2949-2957.
