
FOUNDATION FIRST
For men and postmenopausal women with osteoporosis at high risk for fracture
YOUR BLUEPRINT FOR BONE REBUILDING
TYMLOS works to address the architecture of bone—helping to improve bone strength.1*
*According to preclinical animal studies.1
REBALANCE
TYMLOS binds to and acts as an agonist on the parathyroid hormone 1 (PTH1) receptor to rebalance the body’s natural bone remodeling cycle.1
REMOVE
Osteocytes detect damage and signal osteoclasts to remove old bone.3

MODEST RESORPTION1
REBUILD
RAPID, SUSTAINED BONE FORMATION1
For illustrative purposes only. Not intended to imply clinical efficacy.
Please scroll down for bone marker data.
RAPID, SUSTAINED BONE FORMATION IN MEN AND POSTMENOPAUSAL WOMEN1
Greater increase of bone formation over bone resorption markers was sustained for the duration of therapy in men and postmenopausal women.1,4,5
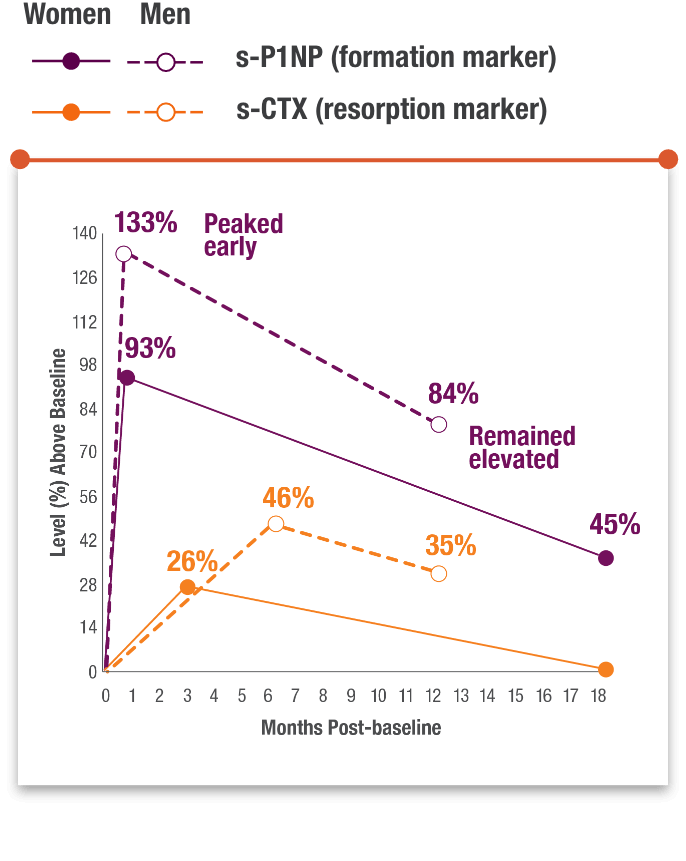
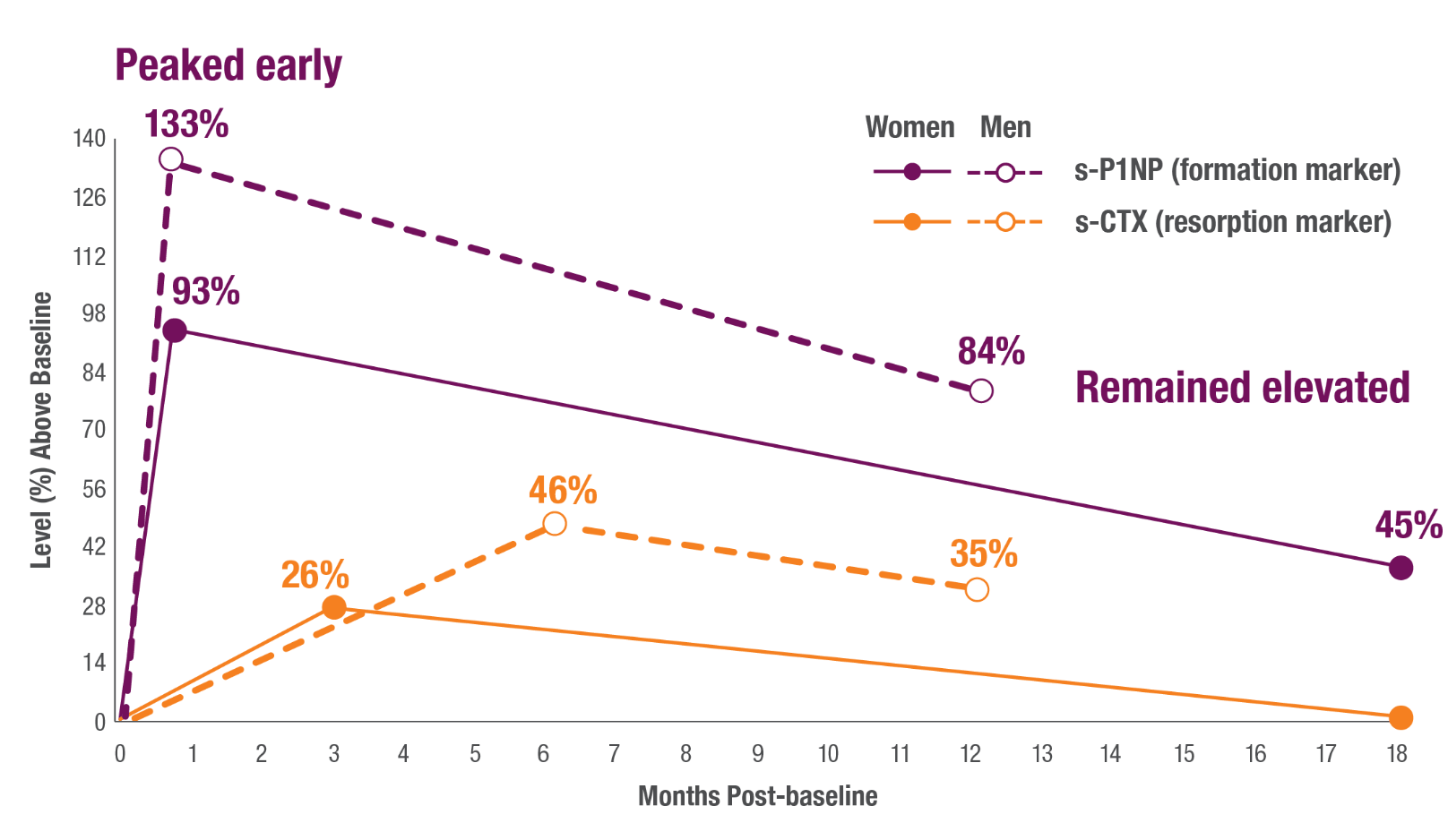
Number of participants evaluated
Postmenopausal women: TYMLOS group: n=189; Placebo group: n=184.
Men: TYMLOS group: n=149; Placebo group: n=79.
s-CTX=serum carboxy-terminal cross-linked telopeptide of type 1 collagen; s-P1NP=serum procollagen type 1 N-terminal propeptide.
TYMLOS quickly shifts the balance during bone remodeling to favor bone formation.1
In preclinical animal studies,
TYMLOS HELPS BUILD BONE1
TYMLOS, a PTHrP(1-34) analog, affected both aspects of bone strength, including BMD and quality.1
Watch how TYMLOS selectively binds to the PTH1 receptor to rebalance the body’s natural bone remodeling cycle.


TYMLOS QUICKLY SHIFTS THE BALANCE DURING BONE REMODELING TO FAVOR BONE FORMATION.1
Watch Dr. David Dempster discuss TYMLOS as a treatment option for both postmenopausal women and men that rebuilds bone.
BONE FORMATION ON THE SURFACE7
TYMLOS RAPIDLY INCREASED MINERALIZING SURFACE AT 3 MONTHS7*
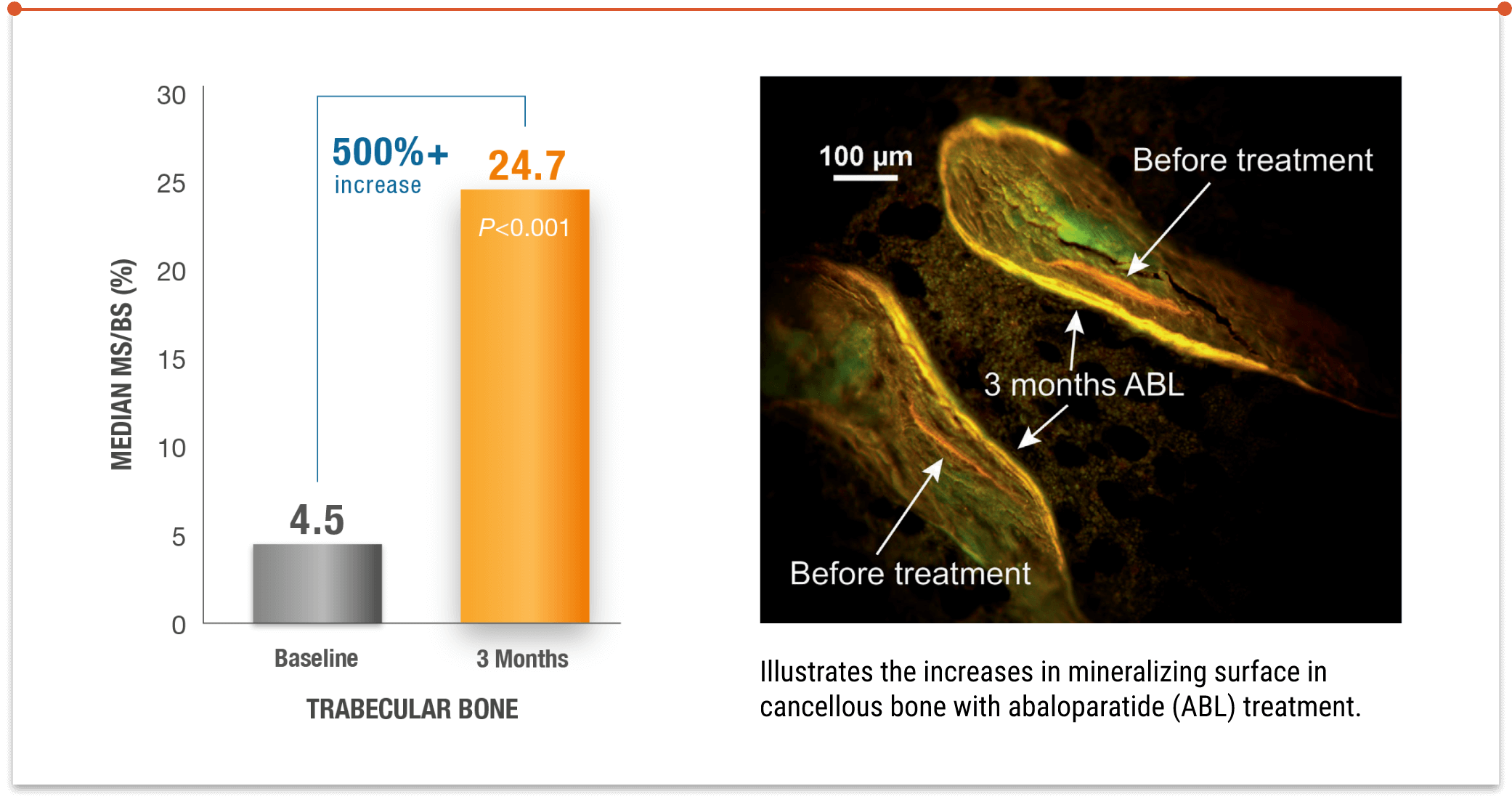
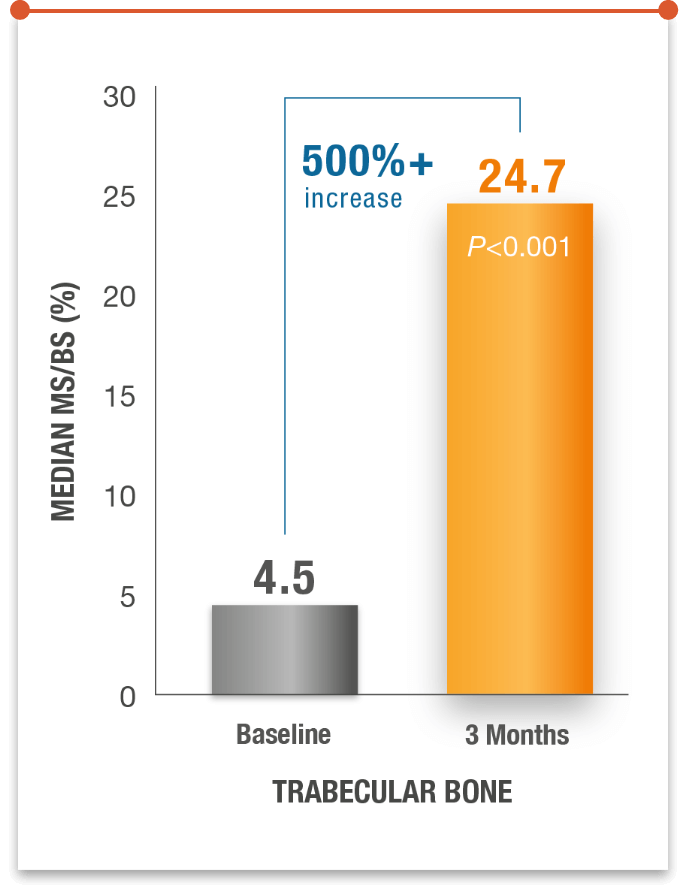
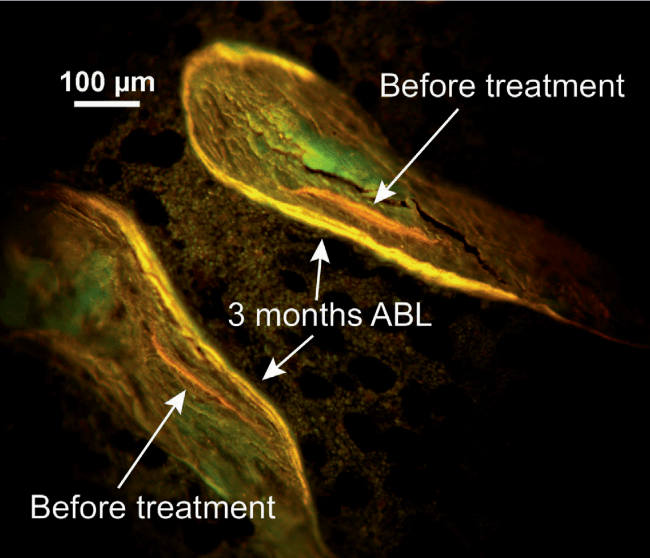
Illustrates the increases in mineralizing surface in cancellous bone with abaloparatide (ABL) treatment.
No new safety signals were seen. Adverse events were similar to those seen in the ACTIVE trial.1,7
MS/BS=mineralizing surface per unit of bone surface.
*3 months is the amount of time it takes bones to form and mineralize in healthy adults.7-9
Should bone density and fracture history guide treatment sequence?
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
IMPORTANT SAFETY INFORMATION
Contraindications: TYMLOS is contraindicated in patients with a history of systemic hypersensitivity to abaloparatide or to any component of the product formulation. Reactions have included anaphylaxis, dyspnea, and urticaria.
Risk of Osteosarcoma: It is unknown whether TYMLOS will cause osteosarcoma in humans. Osteosarcoma has been reported in patients treated with a PTH-analog in the post marketing setting; however, an increased risk of osteosarcoma has not been observed in observational studies in humans. There are limited data assessing the risk of osteosarcoma beyond 2 years of TYMLOS use. Avoid use of TYMLOS for patients at an increased baseline risk for osteosarcoma including patients with open epiphysis (pediatric and young adult patients); metabolic bone diseases other than osteoporosis, including Paget’s disease of the bone; bone metastases or a history of skeletal malignancies; prior external beam or implant radiation therapy involving the skeleton; or hereditary disorders predisposing to osteosarcoma.
Orthostatic Hypotension: Orthostatic hypotension may occur with TYMLOS, typically within 4 hours of injection. Associated symptoms may include dizziness, palpitations, tachycardia, or nausea, and may resolve by having the patient lie down. For the first several doses, TYMLOS should be administered where the patient can sit or lie down if necessary.
Hypercalcemia: TYMLOS may cause hypercalcemia. TYMLOS is not recommended in patients with pre-existing hypercalcemia or in patients who have an underlying hypercalcemic disorder, such as primary hyperparathyroidism, because of the possibility of exacerbating hypercalcemia.
Hypercalciuria and Urolithiasis: TYMLOS may cause hypercalciuria. It is unknown whether TYMLOS may exacerbate urolithiasis in patients with active or a history of urolithiasis. If active urolithiasis or pre-existing hypercalciuria is suspected, measurement of urinary calcium excretion should be considered.
Pregnancy and Lactation: TYMLOS is not indicated for use in females of reproductive potential.
Adverse Reactions:
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in postmenopausal women with osteoporosis are hypercalciuria (11%), dizziness (10%), nausea (8%), headache (8%), palpitations (5%), fatigue (3%), upper abdominal pain (3%), and vertigo (2%).
- The most common adverse reactions (incidence ≥2%) reported with TYMLOS in men with osteoporosis are injection site erythema (13%), dizziness (9%), arthralgia (7%), injection site swelling (7%), injection site pain (6%), contusion (3%), abdominal distention (3%), diarrhea (3%), nausea (3%), abdominal pain (2%), and bone pain (2%).
- treatment of postmenopausal women with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy. In postmenopausal women with osteoporosis, TYMLOS reduces the risk of vertebral fractures and nonvertebral fractures.
- treatment to increase bone density in men with osteoporosis at high risk for fracture (defined as a history of osteoporotic fracture or multiple risk factors for fracture), or patients who have failed or are intolerant to other available osteoporosis therapy.
References: 1. TYMLOS. Prescribing information. Radius Health, Inc. 2. Hattersley G, Dean T, Corbin BA, et al. Binding selectivity of abaloparatide for PTH-type-1 receptor conformations and effects on downstream signaling. Endocrinology. 2016;157(1):141-149. 3. Šromová V, Sobola D, Kaspar P. A brief review of bone cell function and importance. Cells. 2023;12(21):2576. doi: 10.3390/cells12212576. 4. Miller PD, Hattersley G, Riis BJ, et al. Effect of abaloparatide vs placebo on new vertebral fractures in postmenopausal women with osteoporosis: a randomized clinical trial. JAMA. 2016;316(7):722-733. 5. Czerwinski E, Cardona J, Plebanski R, et al. The efficacy and safety of abaloparatide-SC in men with osteoporosis: A randomized clinical trial. J Bone Miner Res. 2022;37(12):2435-2442. doi:10.1002/jbmr.4719. 6. Felsenberg D, Boonen S. The bone quality framework: determinants of bone strength and their interrelationships, and implications for osteoporosis management. Clin Ther. 2005 Jan;27(1):1-11. doi: 10.1016/j.clinthera.2004.12.020. 7. Dempster DW, Zhou H, Rao SD, et al. Early effects of abaloparatide on bone formation and resorption indices in postmenopausal women with osteoporosis. J Bone Miner Res. 2021;36(4):644-653. 8. Baron R, Hesse E. Update on bone anabolics in osteoporosis treatment: rationale, current status, and perspectives. J Clin Endocrinol Metab. 2012;97(2):311-325. 9. Eriksen EF. Cellular mechanisms of bone remodeling. Rev Endocr Metab Disord. 2010;11(4):219-227.
